Absolute Dating/Radioactive Dating
New Rochelle High School, Westchester County
Summer Research Program for Science Teachers
August 2009
OBJECTIVE:
Students will investigate radioactive decay, and understand it in terms of half-lives. They will be able to explain, understand and graph the relationships between the number of radioactive atoms versus stable atoms.
MATERIALS:
Plastic cups
Paper plates
A
bag of Skittles for each group
VOCABULARY:
RADIOACTIVE DEACY
DAUGHTER ISOTOPE
HALF LIFE
RADIO ISOTOPE
INTRODUCTION:
UNSTABLE atoms (with excess energy)
undergo spontaneous (look it up) breakdown into more STABLE (lower energy)
forms.
These unstable atoms are called RADIO ISOTOPES, and they
break down by the process of RADIOACTIVE DECAY.
The RADIO in these words comes from the
fact that the energy they lose as they become stable is given off as RADIATION,
and the atoms are said to be RADIOACTIVE.
The STABLE DECAY PRODUCT (the new, lower energy
atoms that result from the decay) is called DAUGHTER ELEMENTS.
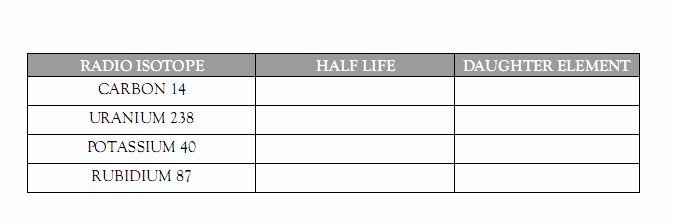
If a certain radioactive element, say
Potassium 40 (written K40), is incorporated into a crystal of
K-Feldspar, it will decay, over time, to the stable element Argon 40 (written Ar40).
When we find the crystal and measure the amount of
the radio isotope K40 and the daughter element Ar40, we
know that if there’s a lot of K40 and not much Ar40 that
the sample has not been around long enough for much K40 to have
decayed - the sample is young!
On the other hand, if there isn’t much K and a lot of Ar we know the crystal is old there must have been a lot of time for all that K40 to decay! Isotopes of various elements decay at different rates, but they all follow a similar pattern of decay. The rate of decay is measured in HALF LIVES - the time it takes for half the atoms in a sample to decay (we have no way of knowing exactly which atoms will decay, but we know half of them will). For example, if there are 100 atoms of K40 in a sample, after one half life there will be only 50 K40‘s left. After the second half life, half of the remaining 50 K40 atoms will decay, leaving 25 K40‘s. This continues on and on until the sample is nearly gone.
The half lives of several radio isotopes are given in your Reference Tables. Look them up and fill in the table below:
In this lab, we’re going to shake and spill Skittles to
model half life.
The
Skittles that land S - up we’ll consider to be RADIOACTIVE, and the S - down
Skittles are a safe stable decay product.
YOU
DO NOT WANT TO EAT RADIOACTIVE SKITTLES - SO NEVER EAT AN S- UP SKITTLE!
Don’t eat any Skittles until you know what you’re doing
with them either.
PROCEDURE:
1. Get a cup of 60 (or so) skittles and a plate to spill them out on. Assume that at one point all the Skittles were S-up on the plate. This represents a sample of 100% radioactive isotope and I’ve entered that data on the data table below.
2. Gently shake the cup of Skittles (make sure we have no flying Skittles) and gently spill them on to the plate. Carefully count and remove all the S- down Skittles, and record your results in the 2nd and 3rd columns on the table below in the 1ST Half Life row. The S - down Skittles you’ve counted are safe to eat now.
3.
Return the radioactive (S - up Skittles) to the
cup, and repeat step 2 above, only this time entering your data in the 2nd
Half Life row.
The counted, recorded and removed S - down Skittles
are OK to eat!
4. Do it again, recording your data in the 3rd half life row. Then do it again, and again, until all of the S - up Skittles are gone.
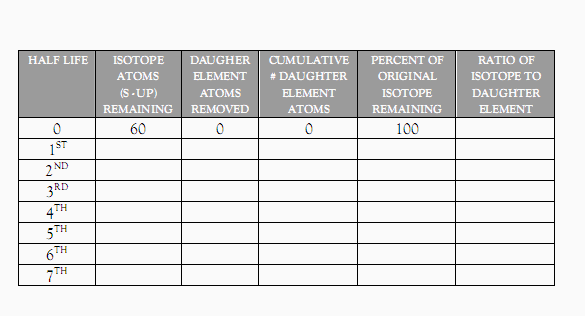
5. Calculate and enter the Cumulative (or total) # of Daughter Elements by adding the together the daughter elements removed in whatever half life you’re on to the sum of all the daughter elements produced in earlier half lives.
6.
Calculate and enter the Percentage of Original
Isotope Remaining by simply dividing the # of Isotope Atoms Remaining by the
total number of original isotopes (here it is 60) and multiplying by 100%
7. Calculate and enter the Ratio of Isotope to Daughter Element by dividing the Isotopes remaining by the Cumulative # of Daughter Elements
8. Prepare 3 neatly labeled graphs, each with the number of half lives on the X-axis:
A) On the 1st graph plot BOTH the Isotope Atoms Remaining and the Cumulative Daughter elements as a function of half life
B) On the second graph, plot the Percent of Isotope remaining as a function of half life.
C) On the third graph, plot the ratio of isotope to daughter element as a function of half life.
DISCUSSION QUESTIONS
1. If the Skittles are C atoms, how many years do 3 half lives represent?
2. If you cut a sample in smaller pieces would that change its half life? Why or why not?
3. About how much of the original U238 that was formed with the earth remains in the planet today?
4. If a sample contains about 40% of its original isotope content, how many half lives old is it?
5. If, in a sample of granite, the ratio of U238 to Pb206 is 1:3, how many half lives old is the sample? How many YEARS old is the sample?
6. Suppose you found a bone of a mastodon which had 6.25% C14 and 93.75% N14. How long ago did this animal die? Does this match with how old mastodons can be?
7. Would C14 be a good choice to determine the age of a trilobite? If not give an alternative isotope (from the ESRT).
The New York State Physical Setting/CORE Curriculum
Standard 4
Students will understand and apply scientific concepts, principles, and theories pertaining to the physical setting.
We will discuss and describe theories about absolute dating methods on Earth.
Students already understand that Geologic History can be reconstructed by observing sequences of rock types and fossils to correlate bedrock at various locations. Today we will evaluate how the rate of nuclear decay, half-life of radioactive isotopes can be used to determine absolute ages of materials within rocks.
Students will use scientific inquiry to pose questions, seek answers, and develop solutions, in order to understand how radioactive atoms decay at observable, constant rates.
Scientific Inquiry, Key Idea 2 &3
Students will develop observations and explanations, using scientific inquiry, in a continuing, and creative process, by performing an experiment to understand the process of nuclear decay and half-lives. The ideas and observations gained during the experiment will be used to construct a graph and interpret and analyze the results, relating them to half-lives.
Standard 6
Students will understand the relationship between mathematics and science by graphing the relationship between the number of trials, compared to the amount of radioactive versus stable atoms.
Key Idea 2
As a class we will be modeling the radioactive decay of isotopes that occurs in some rocks, by working in groups of two, they will shake a beaker with Skittles in it. The ones which end up as “S side up”, will be removed and represent stable decay atoms, and the ones that are “S side down” will represent radioactive atoms. One shake will represent a half-life.