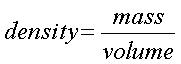 The
formal definition of density is mass per unit volume. Usually the density
is expressed in grams per mL or cc. Mathematically a "per" statement is
translated as a division. cc is a cubic centimeter and is equal to a mL
Therefore,
The
formal definition of density is mass per unit volume. Usually the density
is expressed in grams per mL or cc. Mathematically a "per" statement is
translated as a division. cc is a cubic centimeter and is equal to a mL
Therefore,Density Challenge
Columbia
University’s
Summer Research Program for Science Teachers
August 2008
Subject: Earth Science
Grade Level: 8th and 9th
Unit: Earth and Space
Objective: Students will be able to:
Prior Knowledge: Students should have a good understanding of matter (e.g. atomic structure, compounds, and mixtures). Students should know how to use a digital balance or triple beam balance (if a non-regents class), and how to measure liquids using a graduated cylinder.
Introduction:
Density measures how heavy something is for its size. Liquids of different densities can not be mixed and will separate with the heavier densities at the bottom and the lighter densities at the top. The different objects float at different levels in the liquid depending on their densities. Density is a physical property of matter, as each element and compound has a unique density associated with it. Density defined in a qualitative manner as the measure of the relative "heaviness" of objects with a constant volume.
For example: A rock is obviously more dense than a crumpled piece of paper of the same size.
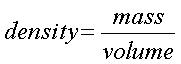 The
formal definition of density is mass per unit volume. Usually the density
is expressed in grams per mL or cc. Mathematically a "per" statement is
translated as a division. cc is a cubic centimeter and is equal to a mL
Therefore,
The
formal definition of density is mass per unit volume. Usually the density
is expressed in grams per mL or cc. Mathematically a "per" statement is
translated as a division. cc is a cubic centimeter and is equal to a mL
Therefore,
Students will be given an opportunity to construct applicable meaning from an inquiry based activity on density. This is a great opportunity to introduce the equations on the front cover of the Earth Science Reference Tables. This understanding concerning density is always evaluated on the Earth Science Regents Exam. The more practice on this subject the better.
Materials:
1. Copies of the Earth Science Reference Tables (cover)
3. Corn syrup (1 liter)
4. Corn oil (1liter)
5. water (1liter)
6. Eight black china markers
7. Chalkboard
8. Chalk
9. Eight digital balances (If a non-regents class triple beam balances may be substituted.)
10. One Whistle
11. One stop watch
12. Eight scientific calculators
Time: 45 minutes
Procedure:
Get two bowling balls one 8lbs and the other 16lbs. Make sure they are the same color. Fill a fish tank with water. Ask the students, showing them both balls, what do you think will happen when I place the balls into the water. Place the balls 16lbs ball first then the 8lbs ball second. Explore with the students explanations for their observations. Then explain When an object is placed in water it will displace its weight in water (Archimede's principle, complement further understanding at http://library.thinkquest.org/27948/archimede.html). Basically the 8 pound ball is displacing 8 pounds of water but since it doesn't take up less volume than 8 pounds of water if floats. Alternatively the 16 pound ball is trying to displace 16 pounds of water but it has less volume than 16 pounds of water and sinks. Therefore we can say the 8 pound ball is less dense than water and the 16 pound ball is more dense than water where density is the mass of the ball divided by its volume.
1. Cooperatively grouped students (e.g. in teams of 4) should find at there stations 4 graduated cylinders marked A, B, C, and D with the black china marker. Cylinder A should be filled with 10 ml of clear corn syrup, cylinder B should be filled with 10 ml of clear corn oil, cylinder C should be filled with 10 ml of water and cylinder D should be left empty. They should have one digital balance at each their station. The setup for each station should be done at least the day before in order for all the liquids to assured of properly settling.
2. Using chalk draw eight tests, each tube divided into 3 sections. I have used precut letter-colored sections of equal sized card board with magnetic backing to be placed in the individual sections. I have also used homemade plastic cylinders with a container of lettered sponge balls to be tossed, in the order of their respective densities, into the tubes. Additionally, another ball may be added for a pinch of fun and labeled D (more work but the results are wonderful to watch). However, they can just as easily draw them into their respective order into the pre-drawn test tubes.
3. Cooperative groups should then be asked to determine the density of each liquid and that when they hear the whistle blow (about 20 minutes) one member from each team will go up to the chalk board with their calculated data, and record their work as quickly as they can. After the member will return to his or her assigned seat before a two minute whistle blows. The first group to correctly assemble the liquids according to their respective densities in the graduated cylinders on the chalk board wins.
4. A summary of students’ findings should be explored, and steps they took to find the correct order of densities related to their experiences with the inquiry based activity, a definition for density should be arrived at, and the official definition of density compared.
Additionally, students may be given, as a follow-up assignment, a web adventure that supports the understanding of density in a real world application. Students may be asked to hand in a summary of their experiences at any of the following links:
http://adventure.howstuffworks.com/sailboat2.htm
For extra credit a book report maybe assigned entitled
By the Great Horn Spoon!
by Sid Fleischman; illustrated by Eric von Schmidt, Atlantic Monthly
Press/Little, Brown and Co.,
Standard 2
All students will understand and apply scientific concepts, principles, and theories pertaining to the physical setting
Standard 5.1
All students will understand the interrelationships between science and technology and develop a conceptual understanding of nature and process of technology.
Standard 5.4
All students will develop problem-solving, decision making, and inquiry skills reflected by formulating usable questions and hypotheses, planning experiments, conducting systematic observations, interpreting and analyzing data, drawing conclusions and communicating results.