|
Element |
Number of Bonds to other atoms |
Angle of Bond Formed |
|
Carbon
|
4 |
|
|
Oxygen |
2 |
O— │
|
|
Hydrogen |
1 |
H—
|
Molecular Structure
Water-Loving and Water-Hating
Molecular Models
Computer models are frequently used by research scientist to understand the complexity of the 3-dimensional shape of molecules. During this lab you will be constructing your own physical model of several molecules. Remember that each atom of a molecule can form a limited number of bonds and these help to determine the shape of the molecule.
|
Element |
Number of Bonds to other atoms |
Angle of Bond Formed |
|
Carbon
|
4 |
|
|
Oxygen |
2 |
O— │
|
|
Hydrogen |
1 |
H—
|
Procedure
1. You must put together a bag of gum drops for your group. Obtain a baggie and place into it the following: 12 white gum drops to represent hydrogen atoms (H), 4 green gum drops to represent oxygen atoms (O), and three red gum drops to represent carbon atoms (C). You will also need 7 or 8 toothpicks which will be used as bonds between the atoms (gum drops). The toothpicks must be cut in half. Once you have all of your materials, you may begin making your molecular models of the following structures. Remember to try to incorporate the angle of the bond as stated in the above chart.
Water Methyl Alcohol Ethyl Alcohol
H—O H H H
│ │ │ │
H H—C —O H— C— C—O
│ │ │ │ │
H H H H H
Once you have completed your models, use color pencils to draw a representation of each of your molecules. Please make a key to indicate the color that represents each atom.
|
Water |
Methyl Alcohol |
Ethyl Alcohol |
|
|
|
|
Compare your models to each other. List 2 ways in which the models are similar.
List 2 ways that they are different from each other.
2. Look at the oxygen molecule of all three models. It actually has a slightly negative charge since it does not share the electron of its bonds equally. Place a BIG negative sign near the oxygen molecule in your drawing. This simply means that the negatively charged electron of the bond spends more time around the oxygen atom. While at the other end of the molecule there is a slight positive charge. Both alcohol and water are polar molecules because of these opposite charges at either end of their molecular structure. Therefore, if you place water and alcohol together, they behave like each other and mix to form a stable solution.
Now, look at the structural formula of Hexane:
H H H H H H
│ │ │ │ │ │
H— C — C — C — C — C — C— H
│ │ │ │ │ │
H H H H H H
The structure of Hexane is different from the models that you made. List two differences.
Hexane, unlike the alcohol, is not soluble in water. Knowing that alcohol mixes in water and
that hexane does not, predict what would occur if the following molecule (simple fat) was placed into water. Would it mix or not?
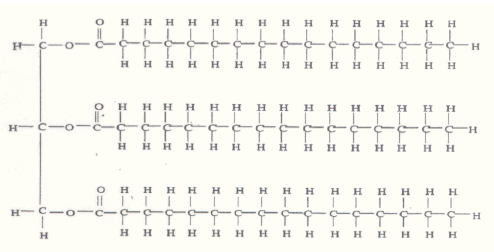
3. You have been provided with the following liquids: Water, Isopropyl Alcohol, Mineral Oil, Ethyl Alcohol, Olive Oil, Acetic Acid (Vinegar), and Corn Oil.
*Obtain 6 test tubes and a test tube rack. Label the tubes 1 to 6 using a “china marker”.
* Decide what mixtures you would like to make. Remember you must only mix two solutions at one time. Record this information in the chart below. You might wish to select mixtures that you are certain that you know the outcome and ones that you do not know the outcome.
*Place 5ml of each solution that you have selected into the appropriate test tube. Remember to rinse-out your graduated cylinder after each usage.
*Carefully place a small piece of wax paper over the tube and invert the tube once or twice
* Record your observation in the chart.
|
Test Tube |
Names of Solutions Tested |
Observations |
Mixable? (yes/no) |
|
1
|
a)
b) |
|
|
|
2
|
a)
b) |
|
|
|
3
|
a)
b) |
|
|
|
4
|
a)
b) |
|
|
|
5
|
a)
b) |
|
|
|
6
|
a)
b) |
|
|
Given your data set, divide the different types of liquids provided in this lab into two groups. Remember that “like dissolves like” and so things that dissolve in each other are similar and should be placed in the same group. If substances do not mix, then they should be placed separate from each other.
|
Group A |
Group B |
|
|
|
4. Look back at the structural formula of water, and ethyl alcohol. Now look at your two groups, are these 2 molecules in the same groups? Remember they all have a slightly negative charge at the oxygen atom and a slight positive charge at the opposite end.
5. Does oil dissolve in water? Do you think that water and oil are similar in structure or different? Would you consider oil water loving or water hating?
6. Dry cleaners use water hating molecules rather than water loving molecules to clean clothes. Explain why using a water hating solutions is effective in removing greasy (oil) stains.
7. Detergents have both a water loving and water
hating part to their molecular structure.
Usually many detergent molecules will surround the structure of an
oil blob. In the space to the right show draw how think this would appear.
|
|
|